How to Make Distilled Water
If you are keen on doing chemical experiments, you will often come across instructions telling you to use distilled water. As a rule, water that has been thoroughly boiled and then carefully poured off so that the dregs or sediment are left behind, will serve the same purpose. If the decanted liquid is again boiled it will be practically as good as distilled water.
But distilled water, and none other, is essential on some occasions, and it is well to know how to make it. Of course, it can be bought reasonably enough at the chemist’s; but the true experimenter likes to depend on his own energies.
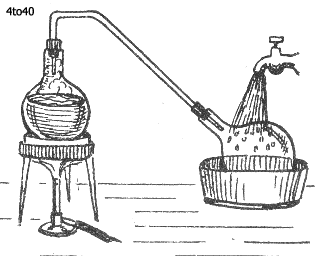
To make distilled water, partially fill a flask with ordinary to tap water, fit a tight cork through which passes a glass tube bent, as shown in the diagram, to join up another flask, also tightly corked.
Stand the first flask on a sand-bath and put it over a lighted Bunsen burner. The sand-bath lessens the chance of the flask being cracked by the heat of the flame.
The heat boils the water and transforms it into vapor, which passes up the tube and down into the second flask. Now, if the second flask is stood in cold water, the hot vapor will be condensed and turned into distilled water. It may be well to say that it takes a long time to make even a cupful of distilled water.
 Kids Portal For Parents India Kids Network
Kids Portal For Parents India Kids Network






