Boiling Point Depends on Pressure
We all know that the boiling point of water is 100 degree Centigrade. But this statement is hardly sufficient. We should add that this is the boiling point at sea-level; in other words, under normal conditions. If we climb a mountain, and so obtain a reduced atmospheric pressure, boiling point will be less than 100 degrees. Conversely, if we descend a mine and so obtain an increased atmospheric pressure, boiling point will be more than 100 degrees.
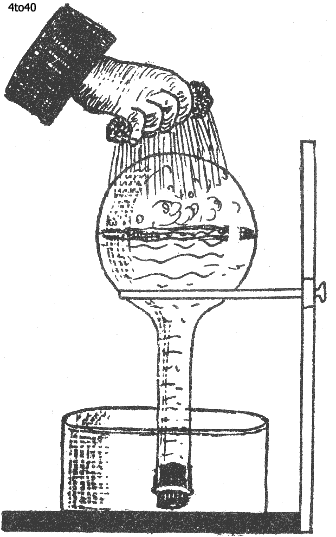
An interesting experiment which illustrates these facts may be performed as follows : Put some water in a globular flask and set it to boil. When boiling has proceeded for some little time and the flask is filled with boiling water and steam, cork it tightly. Remove it from the source of heat, and in a short while boiling will cease. Invert the flask and pour a little cold water over the globular part. At once the water begins to boil again. Why?
The cold water sprinkled over the flask condenses some of the internal vapor. This diminshes the pressure on the internal water,and,because the pressure is less, boiling can occur once more.
 Kids Portal For Parents India Kids Network
Kids Portal For Parents India Kids Network






