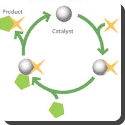
 A catalyst is any substance which causes a chemical reaction without itself undergoing any chemical change. Many industrial processes depend on this property. A simple example is when oxygen and hydrogen combine together quickly in the presence of platinum. Platinum is therefore, the catalyst. It is believed that this is due to the gases being absorbed on the surface of the metal. Magnesium is used as a catalyst for removing unwanted oxides and sulphur during the making of alloys, such as bronze, nickel, or brass.
A catalyst is any substance which causes a chemical reaction without itself undergoing any chemical change. Many industrial processes depend on this property. A simple example is when oxygen and hydrogen combine together quickly in the presence of platinum. Platinum is therefore, the catalyst. It is believed that this is due to the gases being absorbed on the surface of the metal. Magnesium is used as a catalyst for removing unwanted oxides and sulphur during the making of alloys, such as bronze, nickel, or brass.
Some of the most remarkable examples of catalysts are to be found in the human body. These are organic catalysts, or enzymes. At body temperature they bring about chemical reactions, like the burning of sugar. In a laboratory this could be done only at very high temperature which would kill any form of life. Each of the body’s catalysts has its own function. Ptyalin, found in saliva, is concerned in the digestion with converting starch into sugar. Pepsin, produced in the stomach, plays a major part in breaking down the proteins in our food.
 Kids Portal For Parents India Kids Network
Kids Portal For Parents India Kids Network