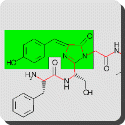
 A chromophore is a group of atoms and electrons (or moiety) i.e. part of organic molecules responsible for its colour. It is an extended delocalised systems of electrons in a compound which gives its colour e.g. chlorophyll’s porphin ring, or an azo dyes benzene ring linked to N=N double bond. When a molecule absorbs certain wavelengths of visible light and transmits or reflects others, the molecule has a colour. A chromophore is a region in a molecule where the energy difference between two different molecular orbitals fall within the range of the visible spectrum. Visible light that hits the chromophore can thus be absorbed by exciting an electron from its ground state into an excited state. In biological molecules that serve to capture or detect light energy, the chromophore is a moiety that causes a conformational change of the molecule when hit by light.
A chromophore is a group of atoms and electrons (or moiety) i.e. part of organic molecules responsible for its colour. It is an extended delocalised systems of electrons in a compound which gives its colour e.g. chlorophyll’s porphin ring, or an azo dyes benzene ring linked to N=N double bond. When a molecule absorbs certain wavelengths of visible light and transmits or reflects others, the molecule has a colour. A chromophore is a region in a molecule where the energy difference between two different molecular orbitals fall within the range of the visible spectrum. Visible light that hits the chromophore can thus be absorbed by exciting an electron from its ground state into an excited state. In biological molecules that serve to capture or detect light energy, the chromophore is a moiety that causes a conformational change of the molecule when hit by light.
Check Also
Kalki 2898 AD: 2024 Indian epic science-fiction dystopian film
Kalki 2898 AD : Movie Name Directed by: Nag Ashwin Starring: Prabhas, Amitabh Bachchan, Kamal …
 Kids Portal For Parents India Kids Network
Kids Portal For Parents India Kids Network